Emetic Agents: From Ipecac to Neuropeptides

The active principle of ipecac root is the alkaloid emetine. In small doses, it serves as an expectorant; in larger quantities, it acts as a potent emetic.
The first chemical warfare agent with emetic properties could well have been the powdered root of the Brazilian plant Ipecac (Carapichea ipecacuanha). Only the prohibitive cost of the raw material prevented its adoption during World War I. Pharmacists who handled the root carelessly would occasionally inhale minute amounts of the dust, resulting in severe irritation of the eyes, nose, and throat, followed by uncontrollable vomiting.[24]
According to American classification, "vomiting agents" include chemical agents such as Adamsite (DM), Diphenylchlorarsine (DA), and Diphenylcyanoarsine (DC). However, their emetic effect is considered secondary to the intense irritation of the upper respiratory tract, which is the primary cause of incapacitation. Their emetic action is not the result of direct stimulation of the brain's vomiting center, and their toxic side effects are so hazardous that they have long been dismissed as viable incapacitating agents. This also applies to other obsolete "emetic gases," such as chloropicrin, phenylimidophosgene, and ethylcarbazole.
Apomorphine
Apomorphine, a semi-synthetic alkaloid, is rightfully considered the most well-known and extensively studied emetic agent. First synthesized in 1869 by heating morphine with sulfuric acid, it derived its name from the Greek prefix "apo-" (meaning "away from" or "derived from"), indicating its origin from morphine. From the latter, apomorphine inherited both analgesic properties and a depressant effect on the respiratory center. It is used in the form of hydrochloride, a white crystalline substance that rapidly turns greenish when exposed to light and air.

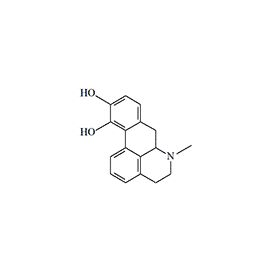
Apomorphine
Structurally, apomorphine belongs to the class of aporphine alkaloids. The progenitor of this class, aporphine, is found in the roots of the water lily (Nymphaea ampla), which grows in regions inhabited by the ancient Maya. According to ethnographer Marlene Dobkin de Rios, it was used by priests in healing practices and lucid dreaming rituals. While history has not preserved specific accounts of the lily root extract's effects on the psyche, it is known that to mitigate the emetic effect of aporphine, priests administered the concoction rectally via enemas. Apomorphine also exhibits psychotomimetic effects in humans; even at therapeutic doses (6–8 mg), it induces hallucinations in one out of every seven patients.[8]
The emetic effect of apomorphine and its derivatives is due to direct action on the D2 dopamine chemoreceptors in the CTZ (chemoreceptor trigger zone). In medicine, apomorphine is used to develop a negative conditioned reflex to alcohol in patients with chronic alcoholism; previously, it was administered when rapid gastric emptying was required in cases of poisoning.
A 2 mg dose of apomorphine—several times lower than the emetic threshold—exhibits sedative and hypnotic effects in humans.[33] At the beginning of the last century, apomorphine was trialed for treating alcoholics, and prior to the discovery of neuroleptics, it was successfully used for the rapid management of agitation in patients with schizophrenia.[34] The American writer William S. Burroughs considered apomorphine the best remedy for treating opiate addiction; he credited the drug with allowing him to remain abstinent for two years, during which he wrote one of his most famous works, Naked Lunch. In recent years, it has been discovered that apomorphine also increases libido in both men and women.
In veterinary practice, apomorphine is still used as an emetic, but exclusively in dogs, as cats tend to fall into a state of frantic excitement following its administration. Occasionally, instead of injections, veterinarians administer apomorphine as eye drops, although this method is considered less reliable.[25]
As an incapacitating agent, apomorphine is entirely unsuitable for aerosol application, as it degrades very rapidly when exposed to light and air. Furthermore, when inhaling the aerosol, an emetic response only occurs at a relatively high concentration of 1000 mg·min/m3. This is far below the effectiveness of modern incapacitants, which exert their effects at concentrations one to two orders of magnitude lower. Even chloropicrin—the "vomiting gas" of the World War I era—induces vomiting at concentrations 10 to 20 times lower.
Therefore, for temporary incapacitation, the only suitable method of administering apomorphine is considered to be by injection. The incapacitating dose of apomorphine via injection is 0.08 mg/kg; in other words, only 5–7 mg is sufficient to induce a bout of agonizing vomiting in a human.
 |
 |
The Palmer Cap-Chur Model 60 pistol (left). William Conner with a Cap-Chur gun for firing apomorphine darts (center). A volunteer is positioned behind glass with a firing port at hip level (right). Photos from the Daily Oklahoman and New York Times (1968).
In 1968, Dr. William Conner of Emory University demonstrated to high-ranking police and military officials the effectiveness of a new riot control tool: pneumatic weapons firing apomorphine-loaded darts. A "flying syringe" and a Cap-Chur pistol, manufactured by the Palmer company and typically used for immobilizing wild animals, were utilized for the demonstration. Doctors and journalists were also in attendance, and the event was covered on a national news program.
According to eyewitnesses, one minute and 15 seconds after the dart struck the student volunteer, the first symptoms appeared: he felt a sudden chill, became glassy-eyed, and his blood pressure dropped. Within two minutes and 45 seconds, he began to feel nauseated. Five minutes after the injection, the man was acutely ill, lying helplessly stretched out on a mattress.[6]
Nevertheless, the injection route also has its drawbacks. The effects of apomorphine are highly inconsistent and individual; as a rule, if vomiting does not occur immediately after administration, subsequent injections prove ineffective. Frequently, increasing the dose leads to a paradoxical antiemetic effect. Despite its relatively high therapeutic index, fatalities have been recorded even after therapeutic doses of apomorphine. In 1962, British Army Captain Billy Clegg-Hill died after being administered emetic injections in an attempt to "cure" his homosexuality.[18]
Agent 926
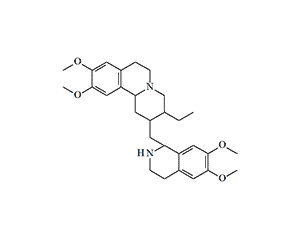
In 1961, the Edgewood Chemical Center contracted the University of Massachusetts to "investigate simple synthetic analogs and other modifications of apomorphine and related alkaloids" and to "synthesize a potent emetic for potential use as an incapacitating agent."[28] In the course of this research, N-substituted norapomorphines were synthesized and studied, some of which surpassed apomorphine's emetic potency by 25–50 times. For further testing as an incapacitant, n-propylnorapomorphine (NPA) was selected. It subsequently became a reference dopamine receptor agonist in experimental medicine and the first "vomiting police gas." Interestingly, the precursor in its synthesis is diacetylmorphine, better known as heroin.[15]
Agent 926
N-n-propylnorapomorphine, ADL 228926, NPA
Following human trials in 1969, N-propylnorapomorphine, designated as Agent 926, was recommended for use as a police "vomiting gas,"[7] though it was never officially adopted for law enforcement. The CIA also showed interest in this compound, experimenting with it under the OFTEN program in the mid-1960s. James S. Ketchum, one of the foremost experts in non-lethal chemical weaponry, expressed concerns that Agent 926 might have been part of the former USSR's chemical arsenal.[26] These fears were not unfounded; indeed, research into emetic and nauseating agents was conducted at GosNIIOKhT (the State Scientific Research Institute of Organic Chemistry and Technology) under the direction of Professor V. Komarov.[17]
 |
R | Name |
ED50 (mg/kg) |
LD50 (mg/kg) |
LD50/ED50 | ICt50 (mg·min/m3) |
|---|---|---|---|---|---|---|
| emesis in dogs, i.v. |
mouse, i.v. |
Safety Index |
emesis in dogs | |||
| H | aporphine | >0.05 | 32 | 640 | N/A | |
| CH3 | apomorphine | 0.012 | 71 | 6,000 | 2000 | |
| C2H5 | N-ethylnorapomorphine (NEA) |
0.00025 | 45 | 180,000 | N/A | |
| C3H7 | N-propylnorapomorphine (NPA) |
0.0005 | 79 | 158,000 | 50 |
ICt50 — the concentration-time product required to induce vomiting in 50% of animals via inhalation.
The next stage in the development of new incapacitants focused on improving the chemical stability of the synthesized compounds. These studies resulted in the production of esters of these emetics, which proved to be far more resistant to atmospheric oxidation than apomorphine.[4]
The effects of these substances on animal behavior were equally unique. While a dose of 0.00075 mg/kg of NPA caused teeth grinding and yawning in monkeys—characteristic symptoms of apomorphine intoxication—higher doses of 10–20 mg/kg induced extraordinary psychological disturbances. These resembled hallucinatory psychosis in humans but were accompanied by an intense drive for self-mutilation, with animals attempting to gnaw off their own limbs. The resulting injuries were so severe that the animals had to be euthanized.[3]
In the late 1970s, the Arthur D. Little Inc. laboratory, continuing the search for a safer replacement for Agent 926, synthesized a 2-hydroxy substituted analog of NPA. This compound was 50 times more potent than apomorphine but did not exhibit adverse effects on the nervous, respiratory, or cardiovascular systems in the event of an overdose. The new agent demonstrated effective emetic action via inhalation.[23]
In 1991, a 2-fluoro substituted analog of NPA was synthesized. Experiments on mice revealed it to be 10 times more potent than its predecessor, Agent 926.[9,10] If further trials confirm such extreme emetic activity in humans, this compound could rightfully be considered the most powerful synthetic emetic known to science.
Clinical Presentation of Poisoning. In terms of emetic activity, Agent 926 is 60–70 times more potent than apomorphine. Following an intravenous dose of 0.00075 mg/kg, a person begins to experience intense nausea within 2–3 minutes, which subsides after 20 minutes. Increasing the dose to 0.001 mg/kg induces a bout of agonizing vomiting in most cases within 2–3 minutes, lasting up to 10 minutes. The median incapacitating dose (ID50) for Agent 926 via intravenous administration was determined to be 0.0012 mg/kg. Data regarding the inhalation toxicity of Agent 926 has not been published in open literature.
Aside from nausea, vomiting, and mild sedation, doses up to 0.001 mg/kg do not cause other noticeable physiological disturbances.[13] Nevertheless, research was terminated in 1970 due to an incident during one of the experiments: after the administration of Agent 926, a test subject suddenly developed a life-threatening cardiac arrhythmia that required emergency resuscitation.[14]
When ingested orally, the toxicity of aporphine emetics in humans is relatively low; the first signs of nephrotoxicity appear at daily doses of 660–1400 mg for apomorphine and over 480 mg for Agent 926.[35]
In addition to their high potency, norapomorphine derivatives possess another property critical for incapacitating agents: their solutions readily penetrate the skin. The minimum transdermal emetic dose for a solution of Agent 926 in benzyl alcohol is 0.015 mg/kg.[3]
Treatment of Poisoning. The use of apomorphine and its derivatives as "vomiting gases" is severely limited by their serious side effects. In some individuals, apomorphine can induce collapse—a sharp drop in blood pressure—which is particularly hazardous for those suffering from cardiovascular disease or atherosclerosis. In patients with gastric ulcers or tuberculosis, intense vomiting may trigger hemorrhaging. Additionally, there is a risk of vomitus entering the respiratory tract, leading to severe aspiration pneumonia.
Antiemetic agents from the D2-antagonist group (such as domperidone or Tigan) can be used to treat poisoning caused by apomorphine and similar emetics. The administration of 5-HT3 antagonists (e.g., ondansetron, granisetron) should be avoided, as they increase the risk of developing hypotension and syncope.[8] In the event of collapse, the victim must be placed in a horizontal position; if the condition does not improve, cardiovascular support medication may be administered.
The antiemetic drug Tigan (trimethobenzamide) has been used quite successfully as an antidote for Agent 926 poisoning.[13]
Substituted Aminotetralins
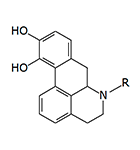
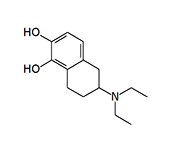
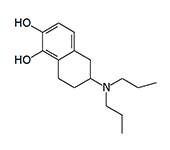
In 1975–1976, J. D. McDermed et al. synthesized 59 new derivatives of 2-aminotetralin. Among them, 5,6-dihydroxy-2-aminotetralins exhibited dopaminergic activity similar to that of apomorphine and induced emesis in dogs following intramuscular administration.[11,12] The structure of these synthesized aminotetralins clearly resembles apomorphine; more precisely, their molecules represent structural fragments of the apomorphine molecule.
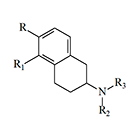 |
Compound | R | R1 | R2 | R3 | EDmin (mg/kg) |
|---|---|---|---|---|---|---|
| 1 | –OCH3 | –OCH3 | –C2H5 | –C2H5 | 0.44 | |
| 2 | –OCH3 | –OCH3 | –CH3 | –n-C3H7 | 0.88 | |
| 3 | –OH | –OH | –C2H5 | –C2H5 | 0.00048 | |
| 4 | –OH | –OH | –n-C3H7 | –n-C3H7 | 0.00057 | |
| 5 | — | –OH | –n-C3H7 | –n-C3H7 | 0.001–0.002 | |
| Apomorphine | 0.026 | |||||
EDmin — the minimum dose of the compound (as a free base) required to induce emesis in dogs.
In addition to the aminotetralins listed above, emetic activity has been observed in N,N-dimethyl-2-aminotetralin, N-methyl-N-propyl-2-aminotetralin, and N,N-dipropyl-2-aminoindane.
The incapacitating concentration (ICt50) for aminotetralin vapors or aerosols is estimated at 50 mg·min/m3, which is 20 times lower than that of apomorphine.[3]
 |
 |
 |
| Derivatives of 5,6-dihydroxy-2-aminotetralin |
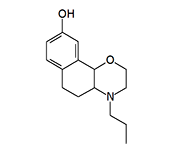
(+)-4-propyl-9-hydroxynaphthoxazine (PHNO) |
|
PHNO (MK-458, HPMC, Naxagolide, Dopazinol). Synthesized in 1982 by chemists at Merck & Co., Inc. as a potential treatment for Parkinson's disease.[1] Like aminotetralins, it belongs to the class of D2-dopaminergic receptor agonists. It is one of the most potent synthetic emetics, inducing vomiting in dogs at a dose of 0.00005 mg/kg (intravenously).[2]
According to Major General N. Antonov of the Soviet Chemical Troops, the ICt50 for PHNO is 5 mg·min/m3.[3] In terms of emetic activity, PHNO is surpassed only by Staphylococcal Enterotoxin B (PG), which induces nausea and vomiting at an ICt50 of 0.5 mg·min/m3, but possesses significantly higher toxicity.
9,10-Dihydrolysergic Acid Derivatives
Hydrogenated ergot alkaloids have long been known in medicine; however, their use is hampered by a distressing side effect: potent emetic action. This is particularly pronounced in 9,10-dihydrogenated derivatives of ergocristine and ergocornine, which are 2 to 4 times more potent than apomorphine as emetics.[30,31] In the early 1970s, the research laboratory at Edgewood Arsenal (Maryland, USA) attempted to synthesize emetic incapacitants based on these compounds.[16]
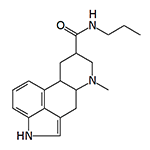
This 9,10-dihydrolysergamide induces vomiting in dogs at a dose of 0.003 mg/kg.
However, even the most active of the resulting compounds were 10–15 times less potent than Agent 926. Interestingly, LSD in high doses (above 0.016 mg/kg) exhibits an emetic effect in nearly one out of every two subjects.[27]
Neuropeptides
Between 1981 and 1983, a study investigating the emetic effects of 17 neuropeptides was conducted under contract with the USAF School of Aerospace Medicine. The research revealed that vasopressin, bombesin, and methionine-enkephalin exhibited the most pronounced emetic effects; these peptides induced brief bouts of vomiting (lasting from ½ to 5 minutes) in dogs following intravenous administration at doses of 0.03–0.08 mg/kg. Slightly less potent were gastrin, substance P, oxytocin, vasoactive intestinal peptide (VIP), and thyrotropin-releasing hormone (TRH).[19,20]
Spatial structure of the Peptide YY molecule
As a rule, vomiting occurred rapidly following the administration of neuropeptides—within 30 seconds—and was typically a single event. Notably, only leucine-enkephalin produced an emetic effect without accompanying side effects. Angiotensin caused thirst, a staggering gait, and dyspnea (shortness of breath) in the animals, while neurotensin induced defecation in addition to vomiting.[21]
The emetic properties of Peptide YY were studied by the Defence Research Establishment Ottawa in 1988. Peptide YY induces vomiting at doses 10 times lower than those of apomorphine. Brief vomiting (no more than three episodes) occurs within 2–3 minutes post-injection.[21,22] Even greater activity has been found in the octapeptide cholecystokinin (CCK-8), which induces vomiting in humans at a dose three times lower than that of Agent 926.[29] Although emetic neuropeptides are occasionally cited as potential incapacitating agents,[32] their practical application remains highly unlikely.
The Unknown Vomiting Gas
In 1969, during the student unrest in Berkeley, California, police extensively used various chemical agents against protesters, including a so-called "vomiting gas." The "Medical Committee for Human Rights" prepared a special "Chemical Warfare Protection Guide for American Students," which described the effects of this "vomiting gas." According to the guide, the substance was colorless, odorless, and deployed in the form of a smoke. Within 2–3 minutes of exposure, victims experienced vomiting and diarrhea, accompanied by severe abdominal cramps. The gas had no lacrimatory (tear-inducing) effect but caused burning pain in the chest. It had a profound demoralizing effect on those affected. Since the substance could penetrate the skin, wearing a gas mask was not only ineffective but also dangerous due to the risk of asphyxiation from one's own vomit. The only way to avoid poisoning was to leave the contaminated area as quickly as possible.[5] The chemical identity of this agent remains unknown.
Conclusion
Despite the existence of compounds with extraordinary potency, such as PHNO or substituted aminotetralins, emetic agents remain a dead end in the development of non-lethal weaponry. The primary obstacle to their practical implementation is an unacceptably narrow therapeutic window: the dose required for reliable incapacitation is dangerously close to the thresholds that trigger critical drops in blood pressure and life-threatening cardiac arrhythmias. In real-world scenarios, where controlling aerosol concentrations is impossible, the high risk of fatalities and severe aspiration complications makes their use unjustifiable. In this regard, modern irritants remain far superior, providing immediate effects with a significantly higher safety index and more predictable physiological outcomes.